Lithiophilic Faceted Cu(100) Surfaces: High Utilization of Host Surface and Cavities for Lithium Metal Anodes - Gu - 2019 - Angewandte Chemie International Edition - Wiley Online Library

Amazon.com : Solo Stove Juniper Firewood for Outdoor Wood Burning Fire Pits and Backyard Fire Pits 100% Natural Juniper Aromatic Firewood Kiln-Dried 6-8 Logs (.5 cu ft) : Patio, Lawn & Garden
Roughening of Copper (100) at Elevated CO Pressure: Cu Adatom and Cluster Formation Enable CO Dissociation

Full atomistic reaction mechanism with kinetics for CO reduction on Cu(100) from ab initio molecular dynamics free-energy calculations at 298 K | PNAS

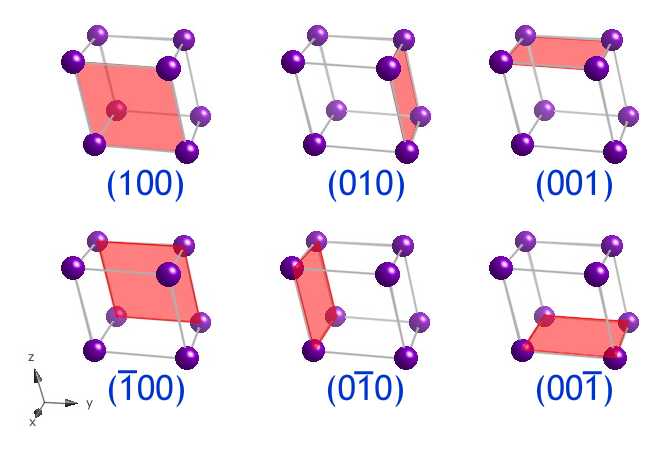
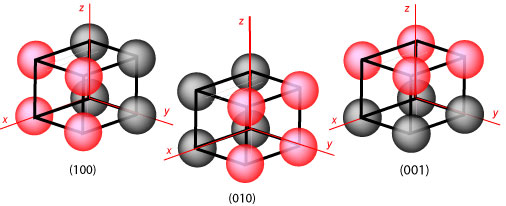
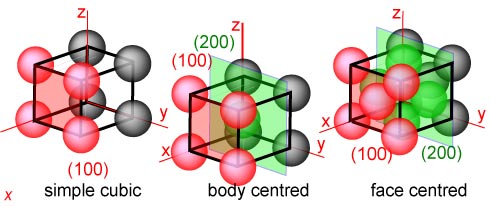
Identification of Cu(100)/Cu(111) Interfaces as Superior Active Sites for CO Dimerization During CO2 Electroreduction | Journal of the American Chemical Society

Mechanism of CO2 conversion to methanol over Cu(110) and Cu(100) surfaces - Dalton Transactions (RSC Publishing)

JFB | Free Full-Text | Synthesis and Biological Activity Evaluations of Green ZnO-Decorated Acid-Activated Bentonite-Mediated Curcumin Extract (ZnO@ CU/BE) as Antioxidant and Antidiabetic Agents

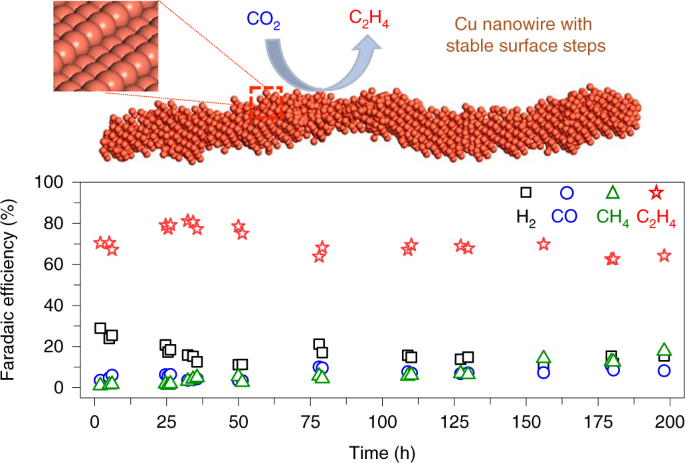
Engineering Cu surfaces for the electrocatalytic conversion of CO2: Controlling selectivity toward oxygenates and hydrocarbons | PNAS

Micrometre-scale single-crystalline borophene on a square-lattice Cu(100) surface | Nature Chemistry
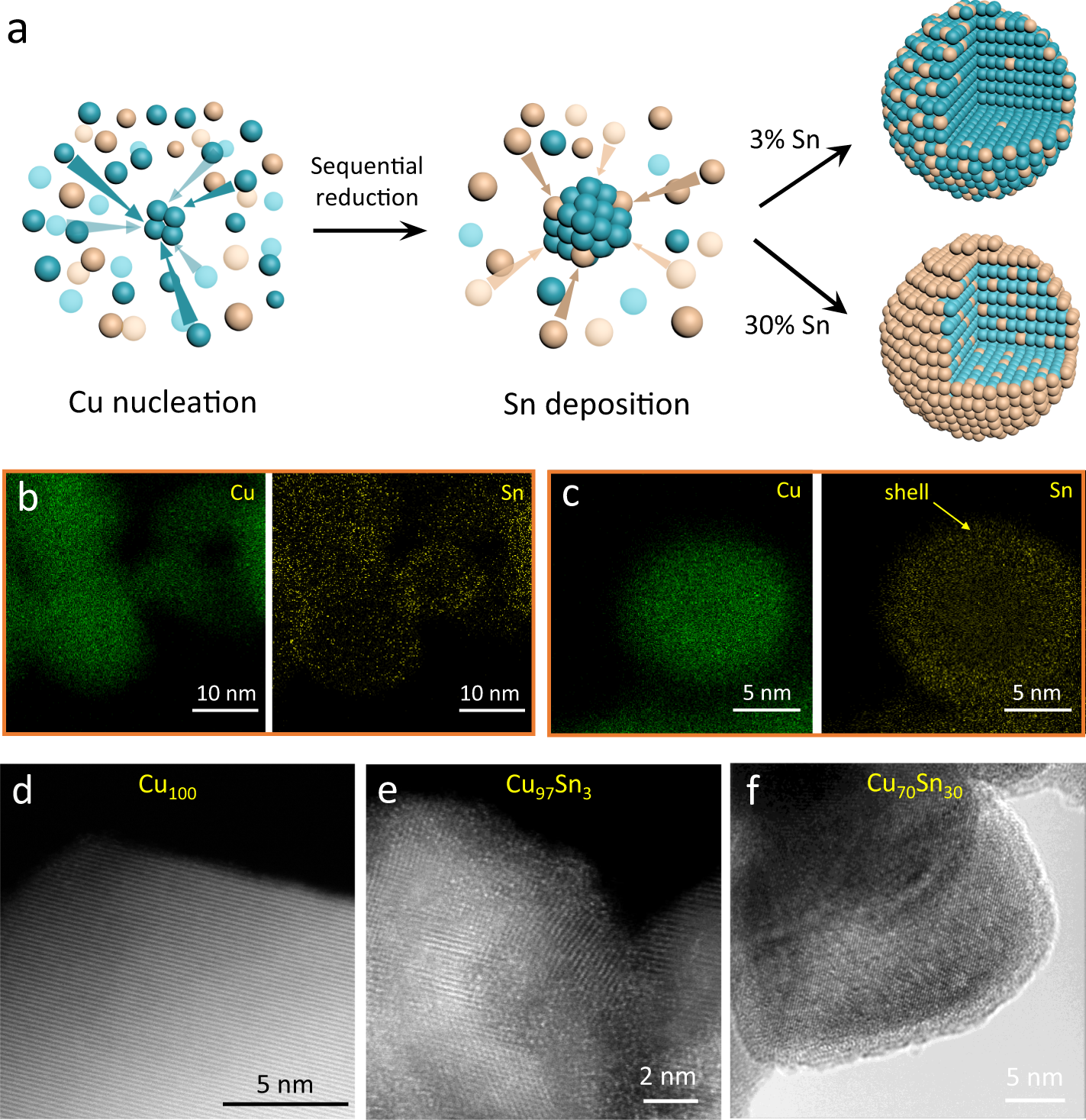
Isolated copper–tin atomic interfaces tuning electrocatalytic CO2 conversion | Nature Communications

Identification of Cu(100)/Cu(111) Interfaces as Superior Active Sites for CO Dimerization During CO2 Electroreduction | Journal of the American Chemical Society