SOLVED: There are several steps to the synthesis of the aluminum potassium alum that can be represented by the overall reaction.2Al(s) + 2KOH(aq) + 4H2SO4(aq) + 24H2O(l) → 2KAl(SO4)212H2O(s) + 3H2(g) +H2O(l)1.

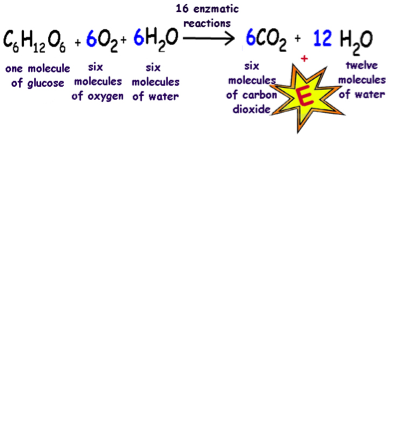
SOLVED: Using the chemical equation.... 2 C6H14 + 19 O2 —-> 12 CO2 + 14 H2O Calculate the number of moles of carbon dioxide produced by the consumption/production of the following. a)
![Aluminum Potassium Sulfate Dodecahydrate [AlK(SO4)2.12H2O] Molecular Weight Calculation - Laboratory Notes Aluminum Potassium Sulfate Dodecahydrate [AlK(SO4)2.12H2O] Molecular Weight Calculation - Laboratory Notes](https://www.laboratorynotes.com/wp-content/uploads/2021/12/aluminum-potassium-sulfate-dodecahydrate-molecular-weight-calculation.jpg)
Aluminum Potassium Sulfate Dodecahydrate [AlK(SO4)2.12H2O] Molecular Weight Calculation - Laboratory Notes

![12" H2O Coil Glass water recycle bong [H2O-18] – Pothead Parent 12" H2O Coil Glass water recycle bong [H2O-18] – Pothead Parent](https://cdn.shopify.com/s/files/1/0605/3917/5125/products/050eed24-12e1-4d34-bb5b-32a8fe97be84.jpg?v=1669868015)