Setara kan persamaan reaksi ini: Al2O3 + H2O ⎯⎯→ Al(OH)3 - Brainly #Backtoschool2019 - Brainly.co.id

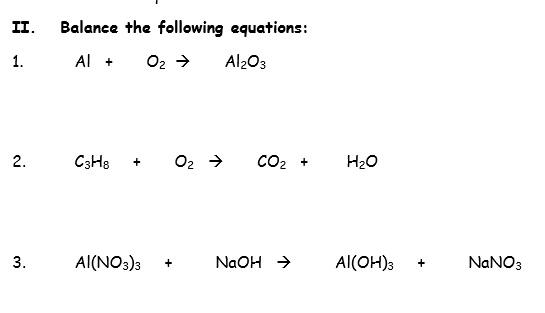
Question: Balance following equations: C3H8 O2 → CO2 H2O Al2(SO3)3 NaOH → Na2SO3 Al(OH)3 Al2O3 - Chem 101 - Stuvia US

Al-alcl3-al(oh)3-al2o3-и посложнее )) (На картинке ) Составьте уравнения :) Спасибо заранее - Школьные Знания.com

0.078 g Al(OH)(3). is dehydrated to Al(2)O(3). The Al(2)O(3) so obtained reacted with 6 milli-equivalent of HCl. The equivalent of AlCl(3) produced during the reaction are:

Complete and balance the following molecular equation. Al(OH)_3(s) + HNO_3(aq) to And write the net ionic equation for it. | Homework.Study.com

4/27/16 Today I will define stoichiometry and calculate mole-mole stoichiometry problems Warm Up Write a balanced equation for the reaction of magnesium. - ppt download

High-Pressure δ-Al(OH)3 and δ-AlOOH Phases and Isostructural Hydroxides/Oxyhydroxides: New Structural Insights from High-Resolution 1H and 27Al NMR | The Journal of Physical Chemistry B