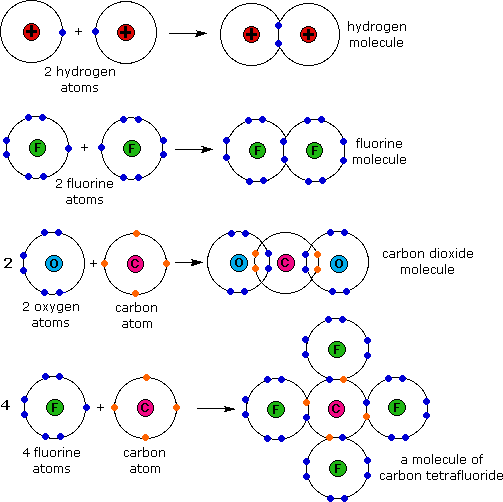
Atomic Counting Answer the following questions by determining the total number of atoms in each formula. 1. H2SO4 2. 3Fe AgNO3 4. Pb(NO3)2 5. Ba(ClO3)2. - ppt download

A compound has the molecular formula X4O6 . If 10 g of X4O6 has 5.72 g X, then the atomic mass of X is :

All Stars ready for Ohio double at Attica and Atomic June 3-4 - All Star Circuit of Champions | 410 Outlaw Sprint Car Series ASCoC - All Star Sprint Series










:max_bytes(150000):strip_icc()/what-is-the-rydberg-formula-604285_final-251d1441e24e44c88aab687409554ed4.png)