How to Balance BaO + H2O = Ba(OH)2 | BaO + H2O = Ba(OH)2 balance| Chemical equation BaO + H2O - YouTube

Balance the following chemical equations 37% also z aher questions to solve 1 NaP03 + H20 2 H2C03 - Science - Chemical Reactions and Equations - 13533925 | Meritnation.com

SOLVED: Which one of the following equations represents neutralization? CO2 +Hz0 + HzCO: BaO + Hz0 + Ba(OH) 2 HBr + NaOH NaBr + HzO 2Li + Br? 2LiBr

SOLVED: Organize the following in order of increasing vapor pressure: H2 02 HzO BaO H2<H2O<O2<BaO BaO<HzO < O2<Hz H2<02<HzO<BaO BaO<H2<02<HzO H2<02<BaO<HzO If the process in question 2 (a reaction has AHS rxn = -
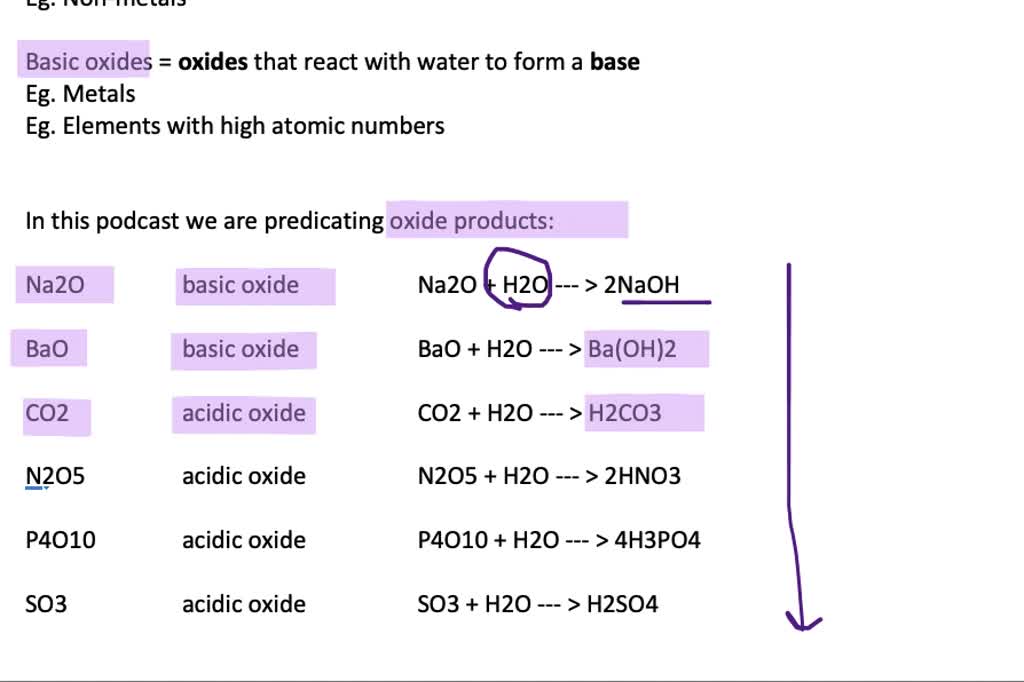
SOLVED:Predict the products of the following oxides with water: Na2 O, BaO, CO2, N2 O5, P4 O10, SO3 . Write an equation for each of the reactions. Specify whether the oxides are