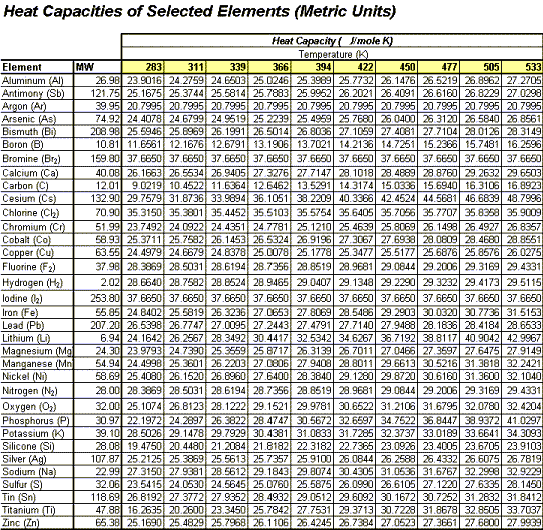
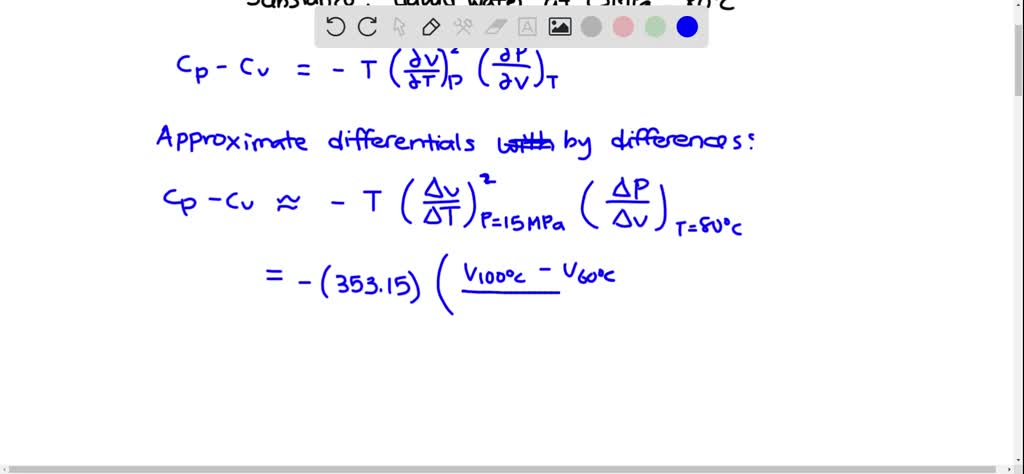
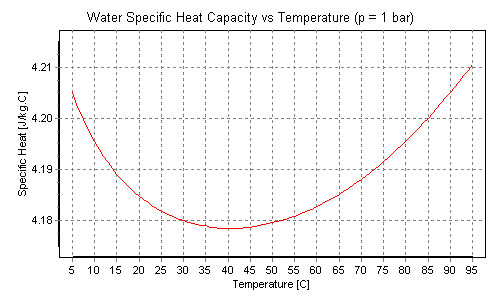
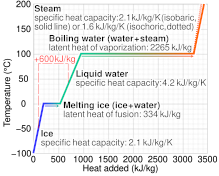
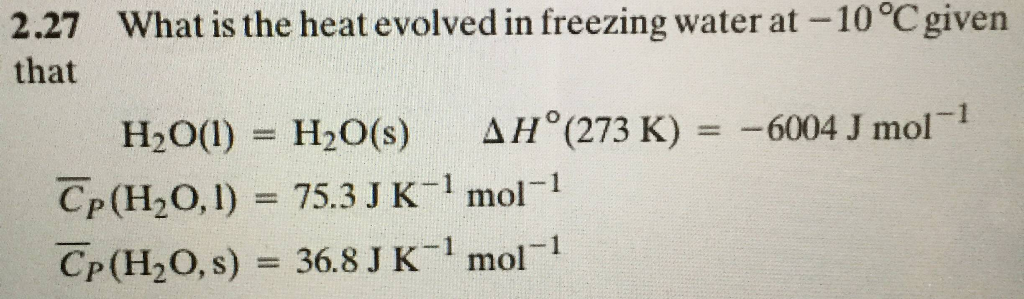The enthalpy change for a reaction at equilibrium is - 20.5 kJ mol ^-1 . Then the entropy change for this equilibrium at 410 K is:

Calcualte the enthalpy change on freezing of 1.0 mole of water at 10.0^(@)C to ice at -10^(@)C. ... - YouTube

The System CaCl2–H2O: Thermodynamic Modeling and Flow Calorimetry Experiments at Elevated Temperatures and Pressures | Journal of Chemical & Engineering Data