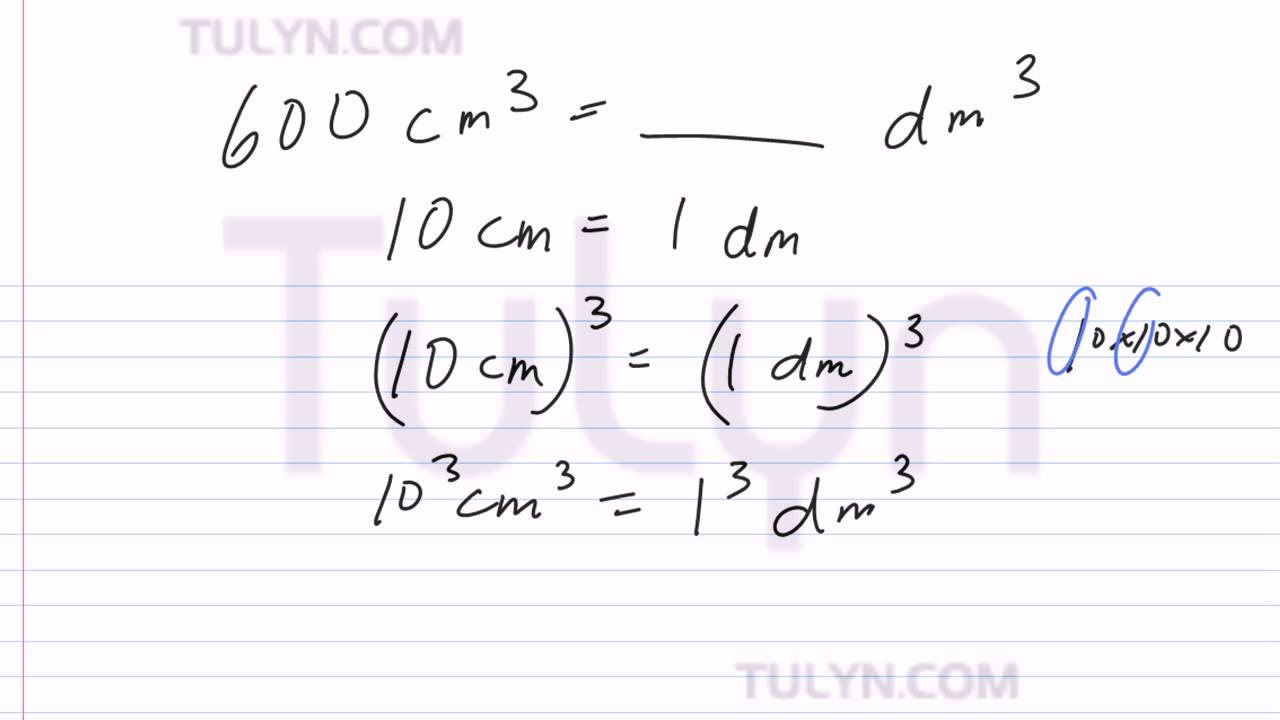Energies | Free Full-Text | A New Control Method for a Bi-Directional Phase-Shift-Controlled DC-DC Converter with an Extended Load Range

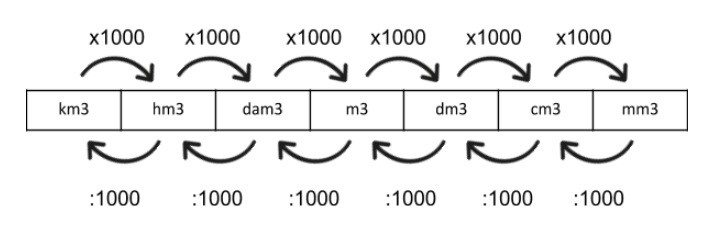
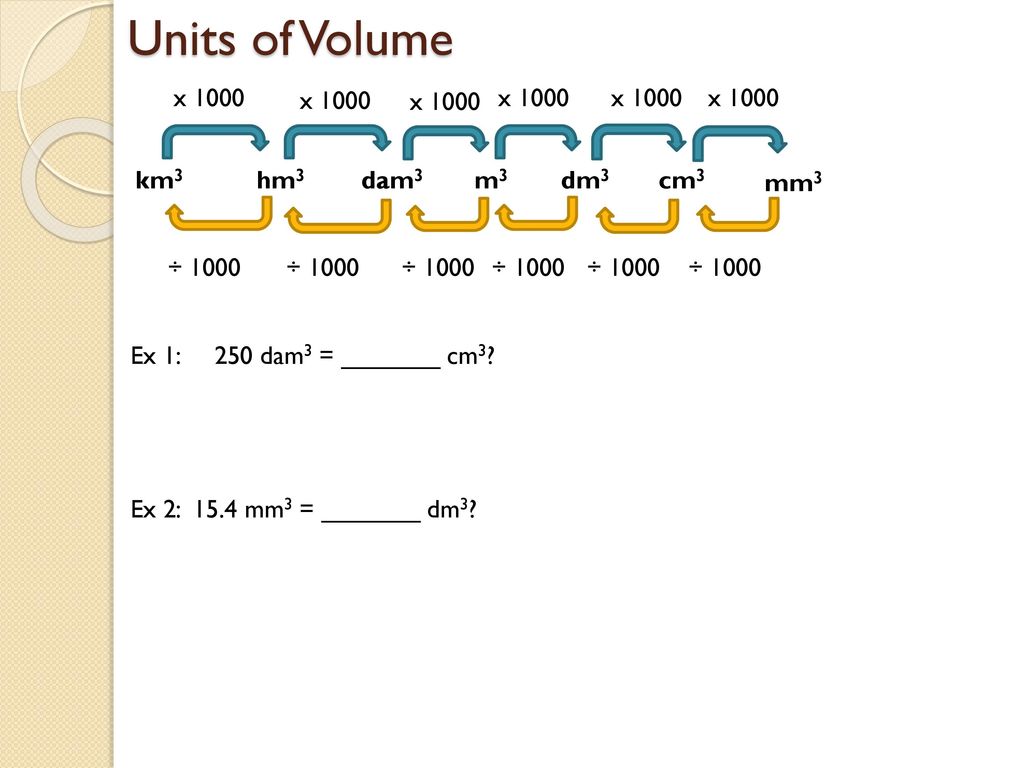
SOLVED: 'supply the missing number Activity: Supply the missing number: 6700 dm3= m3 1 28 dm3= cm3 2 11500 cm3 m3 3 4 4m3 = cm3 6,% 5 8m3 = dm'

SOLVED: Q1 If molarity of Na2CO3is 0.05 M at a volume of 25.00 cm3 while the titre value of HClis 14.00 cm3, calculate the concentration of HCIin molldm3. Equation of the reaction

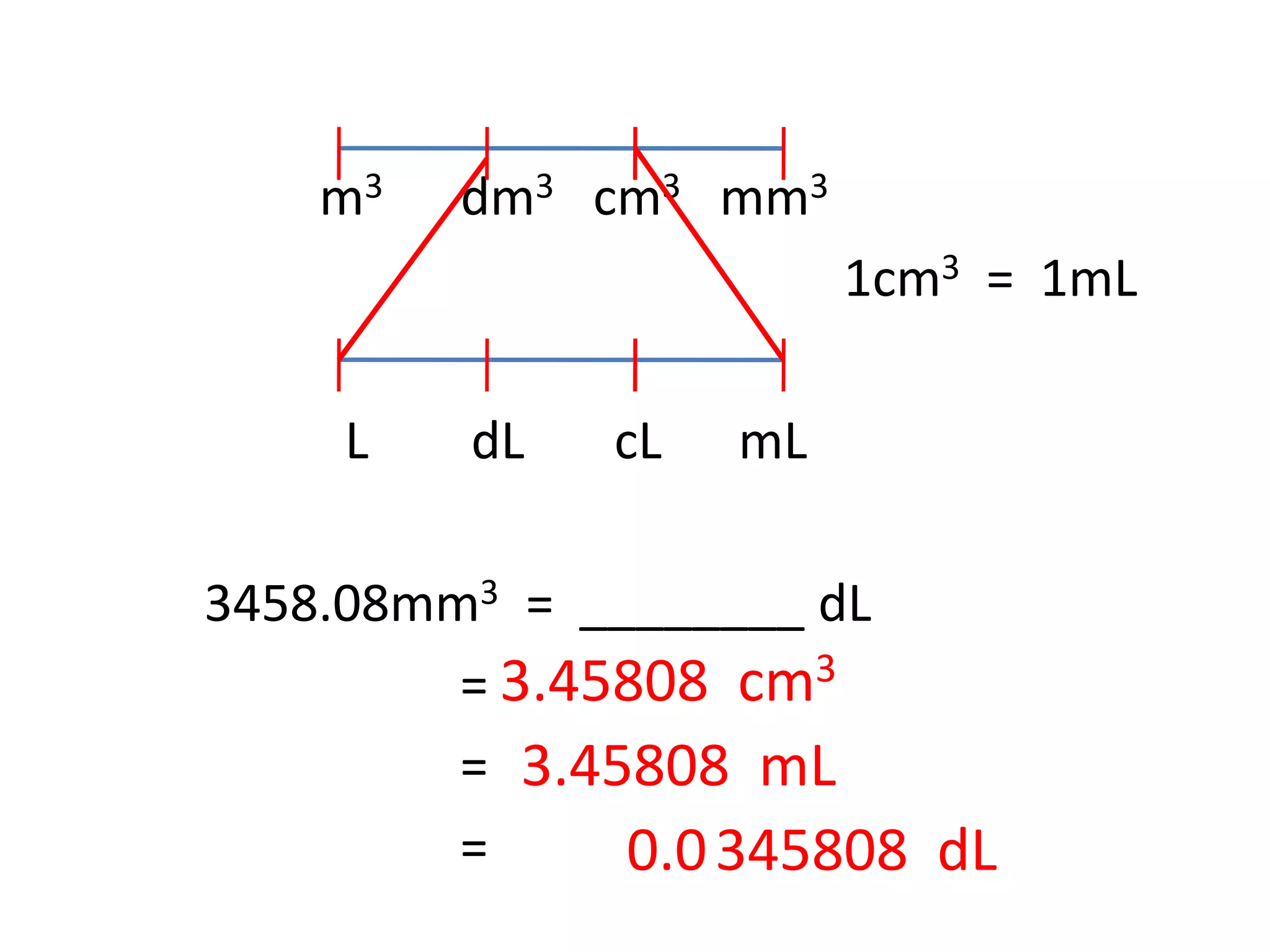
Try This!OOsoFill in the blanks.a 1 litre =b 5 litres =millilitresdm3-cm3- dm3• cm3litresc 10,000 cm3 - Brainly.in