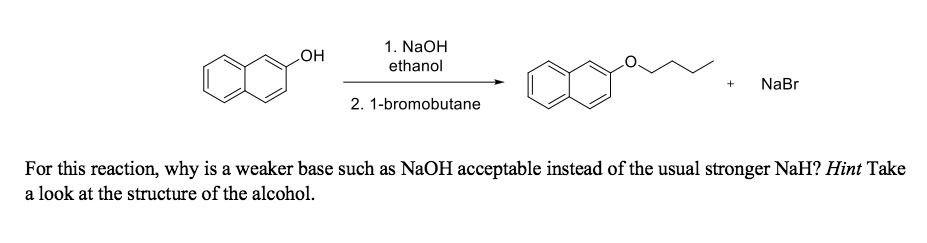
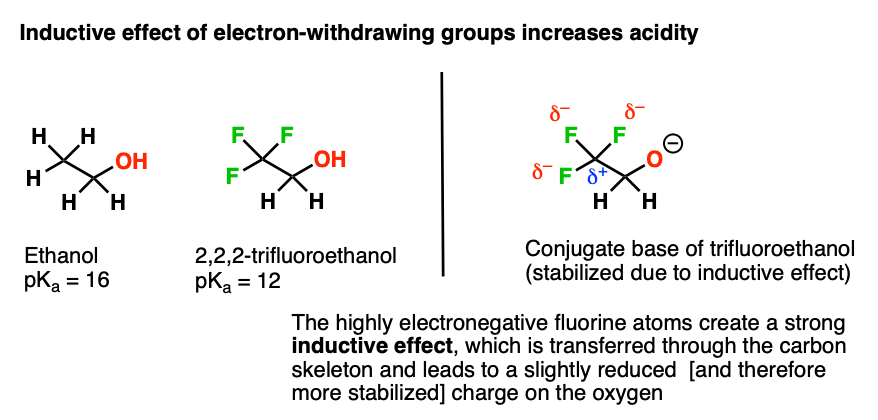
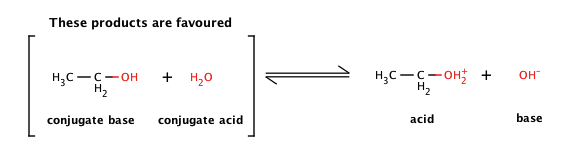
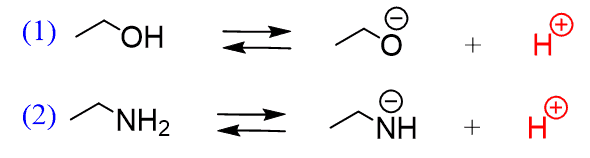
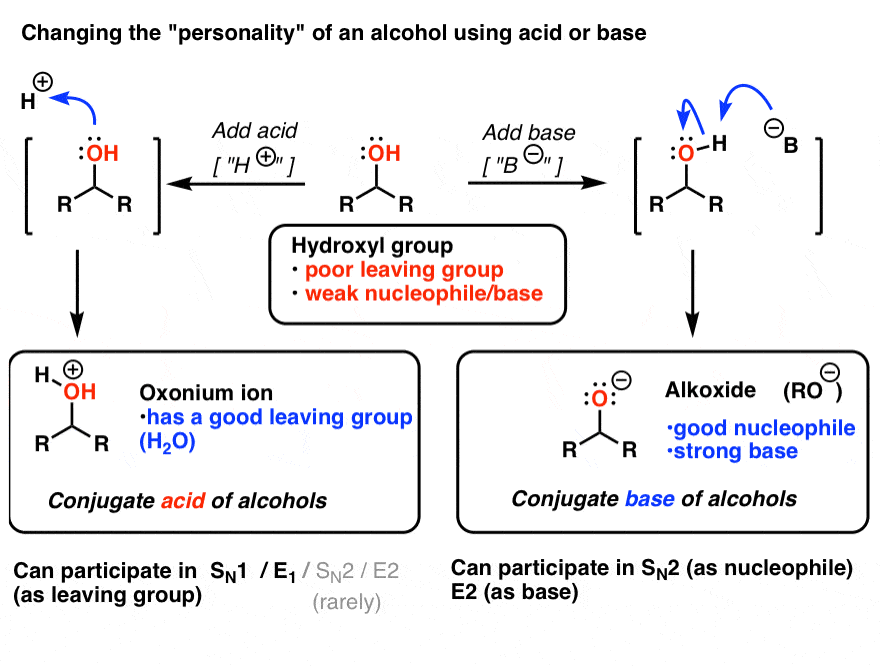
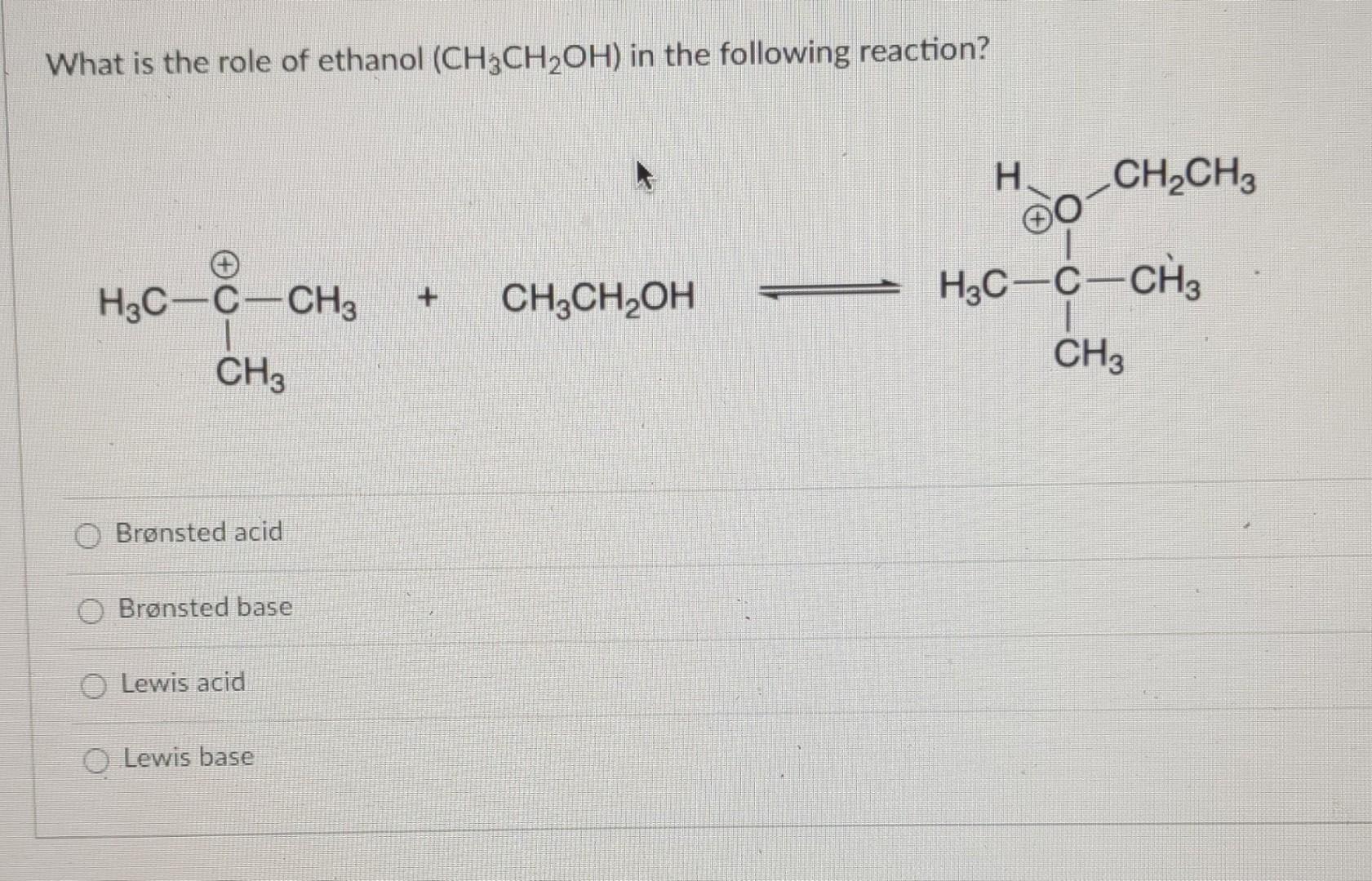
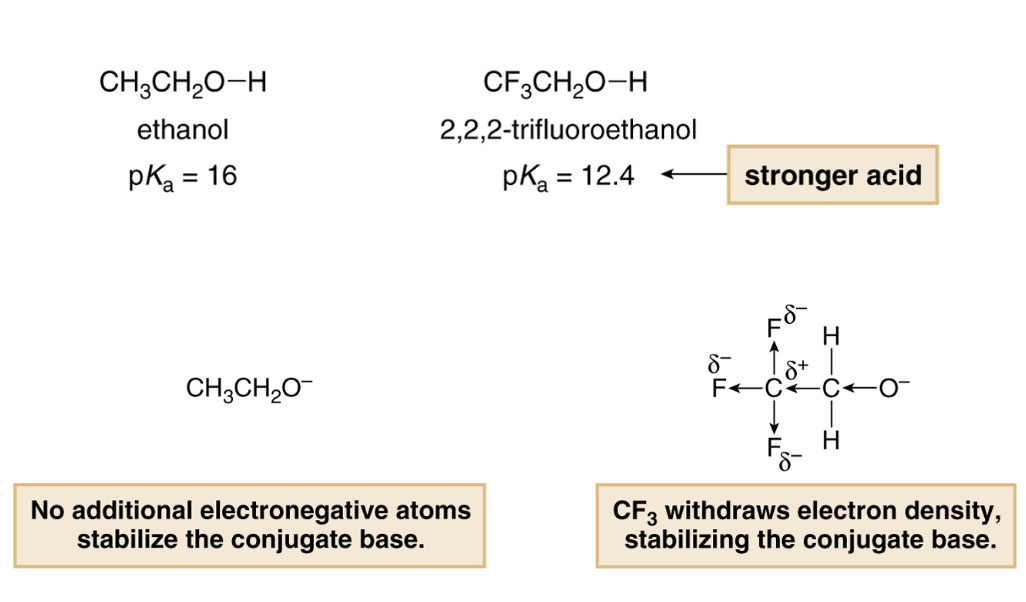
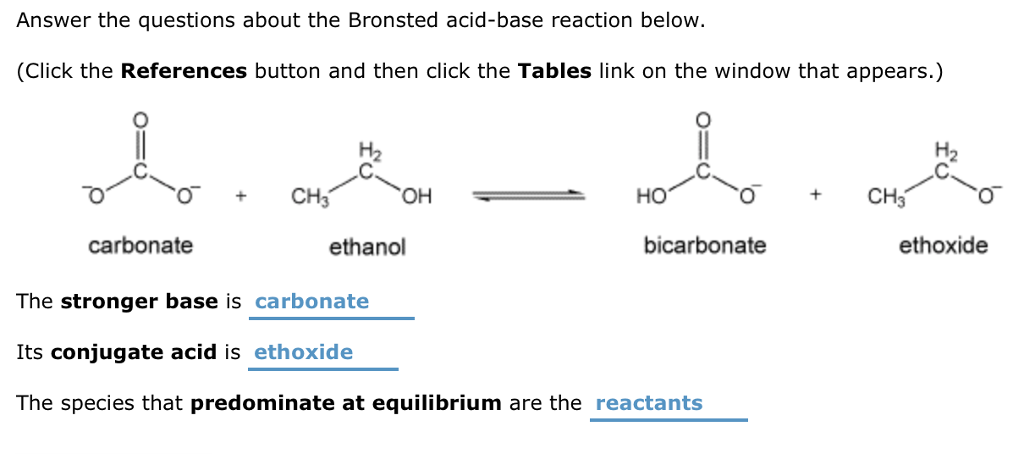
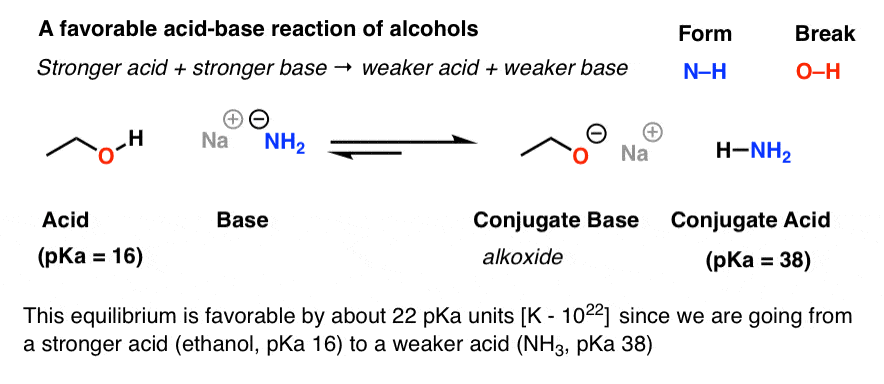1,1,1-Trichloroethanol is an acid more than 1000 times stronger than ethanol, even though both have a conjugate base where the negative charge is on an oxygen. Provide an explanation for this observation.

HOMCOM Portable Tabletop Ventless Bio Ethanol Fireplace with Glass Walls Stainless Steel Arc Base Bronze | Oriental Trading

Ethanol condensation to butanol at high temperatures over a basic heterogeneous catalyst: How relevant is acetaldehyde self-aldolization? - ScienceDirect

Northlight 10.5 in. Bio Ethanol Round Portable Tabletop Fireplace with Silver Base 34808727 - The Home Depot