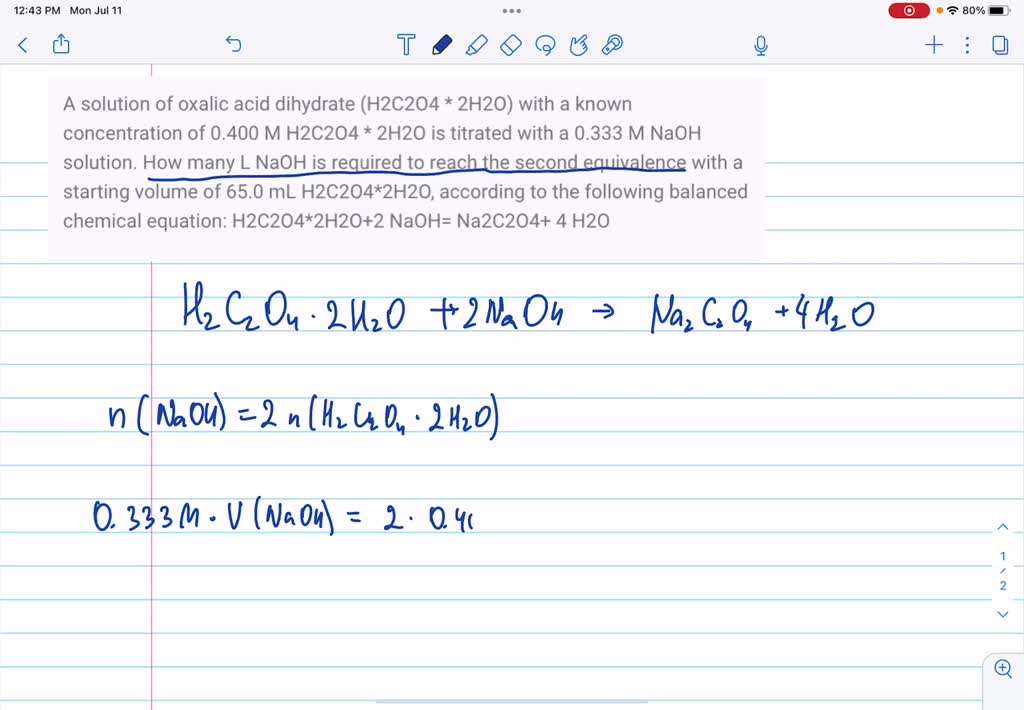
SOLVED: How much oxalic acid (H2C2O4 with 2H2O) is required to prepare 1000ml of solutions of the following respective normalities: a.)0.5000, b.)0.2500, c.)0.200?

Best Price H2c2o4.2H2O 99.6%Min Oxalic Acid for Polishing and Cleaning Chemical - China Oxalic Acid and Oxalic
Calculate the amount of oxalic acid (H2C2O4.2H2O) required to obtain 250 mL of deci-molar solution. - Sarthaks eConnect | Largest Online Education Community

What mass of oxalic acid dihydate, H2C2O4•2H2O, is needed to make a 0.498 M solution of oxalic acid in a 250.0 mL volumetric flask? - Quora
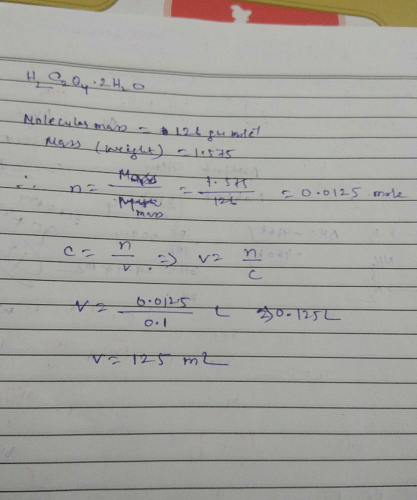
Volume (mL) of H2C2O4. 2H2O solution to prepare 0.10 M from 1.575g of it is ?Correct answer is '125'. Can you explain this answer? | EduRev Chemistry Question
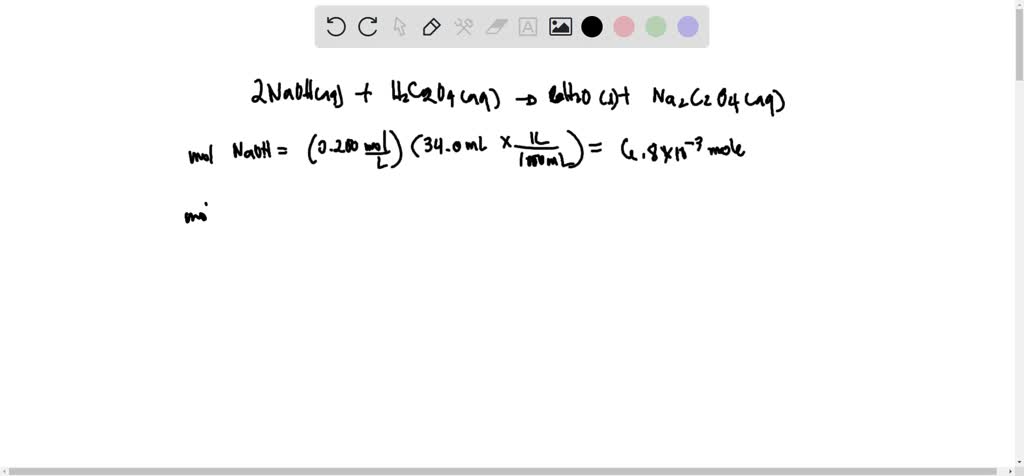
SOLVED: 2 NaOH(aq) + H2C2O4 = 2H2O(s) Na2C2O4(aq) + 4 H2O(l) Calculate the molarity of an oxalic acid solution if it takes 34.0 mL of 0.200 M NaOH solution to consume the

H2c2o4.2h2o High Purity Oxalic Acid 99.6% - Buy Oxalic Acid 99.6%,Oxalic Acid Price,Oxalic Acid Product on Alibaba.com

SOLVED: 116.5 g of oxalic acid dihydrate, H2C2O4 · 2H2O (MM 126.07 g/mol), is used to prepare 350.0 mL of solution 1? What is the molarity of a new solution made by



![Oxalic Acid Dihydrate [H2C2O4 2H2O ] [CAS_6153-56-6] 99.6+% Fine White – Wintersun Oxalic Acid Dihydrate [H2C2O4 2H2O ] [CAS_6153-56-6] 99.6+% Fine White – Wintersun](https://cdn.shopify.com/s/files/1/0724/7981/products/15-005-1_1024x1024.jpg?v=1550179687)





![Oxalic Acid Dihydrate [H2C2O4 2H2O ] [CAS_6153-56-6] 99.6+% Fine White – Wintersun Oxalic Acid Dihydrate [H2C2O4 2H2O ] [CAS_6153-56-6] 99.6+% Fine White – Wintersun](https://cdn.shopify.com/s/files/1/0724/7981/products/15-005-4_1024x1024.jpg?v=1662152063)