Predict which of the ions Cu^+, Sc^3+, Mn^2+, Fe^2+ are coloured in aqueous solution? Give reasons. - Sarthaks eConnect | Largest Online Education Community

SOLVED: The product solubility constant for copper (I) iodide is 1.1X 10-12 When 0.40 M HCN is added to this ionic salt; forming the complex ion, Cu(CN) 2 with a product formation constant
Ch 7.1 Forming Ions. Review… Cations are Groups 1A, 2A, and 3A They have positive charges. Anions are Groups 5A, 6A, and 7A They have negative charges. - ppt download

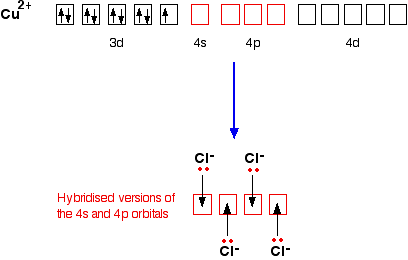
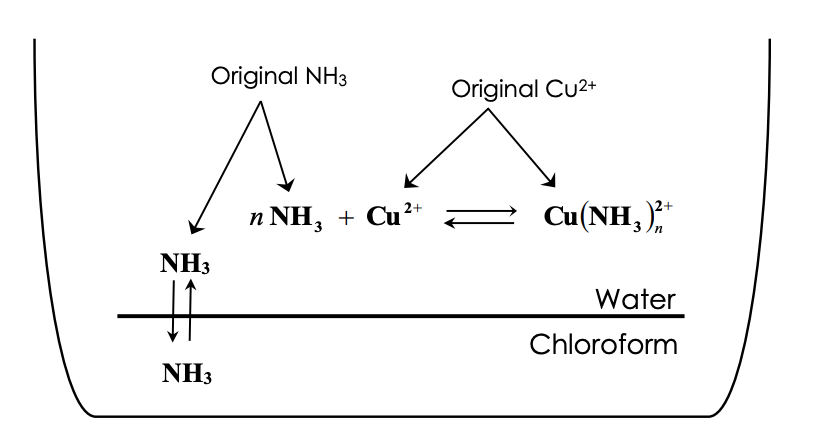
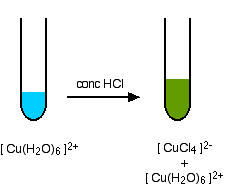
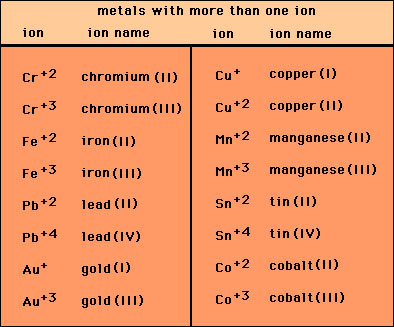
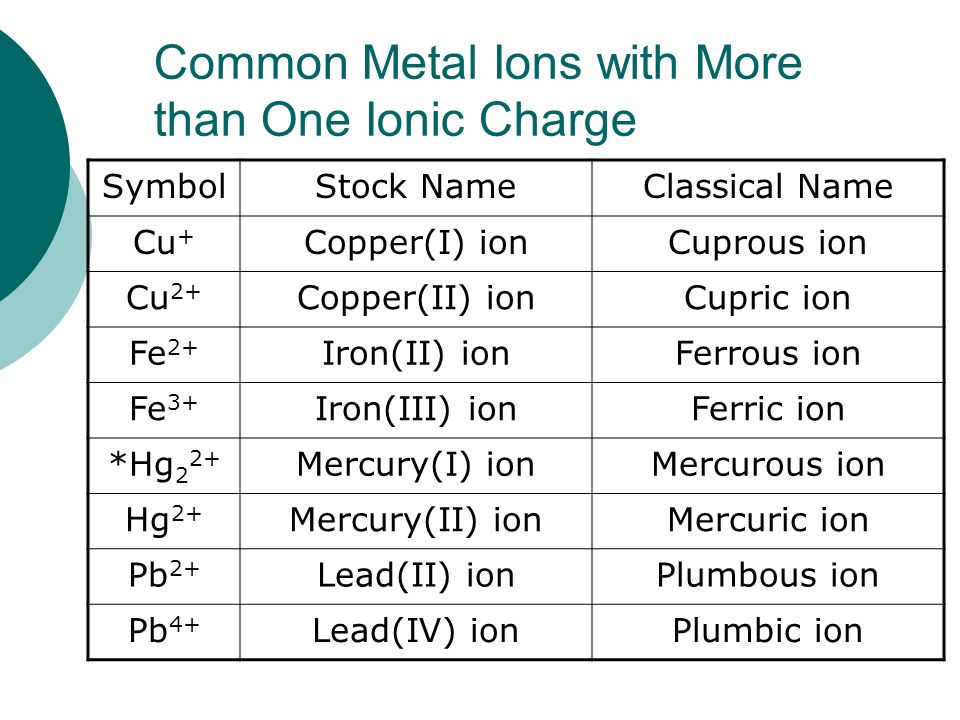
Copper Cu transition metal Chemistry copper(I) Cu+ copper(II) Cu2+ ion complex ions ligand substitution compounds redox chemical reactions principal oxidation states +1 +2 GCE AS A2 IB A level inorganic chemistry revision
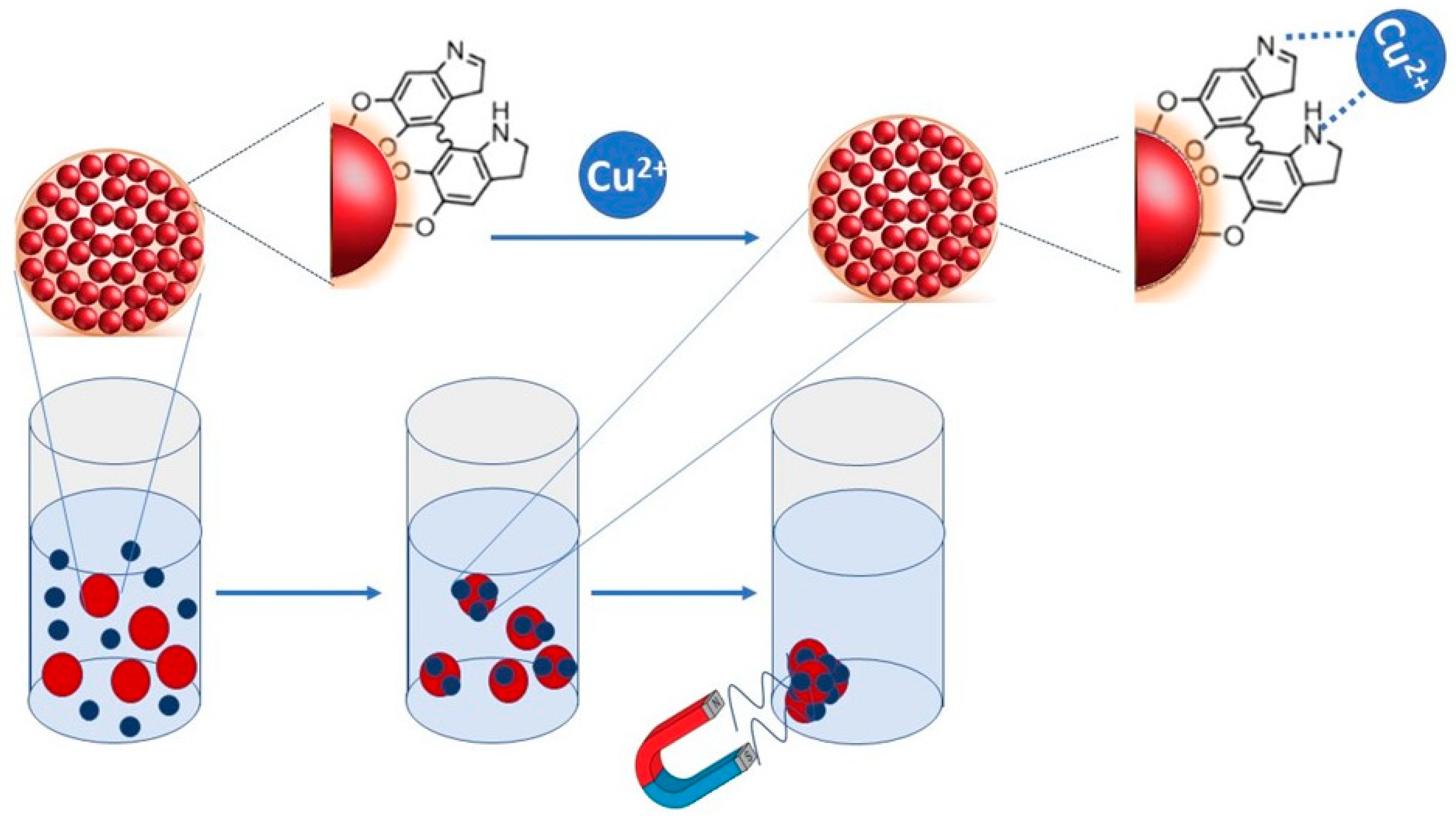
Materials | Free Full-Text | Synthesis and Characterization of SPIONs Encapsulating Polydopamine Nanoparticles and Their Test for Aqueous Cu2+ Ion Removal

Copper Cu transition metal Chemistry copper(I) Cu+ copper(II) Cu2+ ion complex ions ligand substitution compounds redox chemical reactions principal oxidation states +1 +2 GCE AS A2 IB A level inorganic chemistry revision

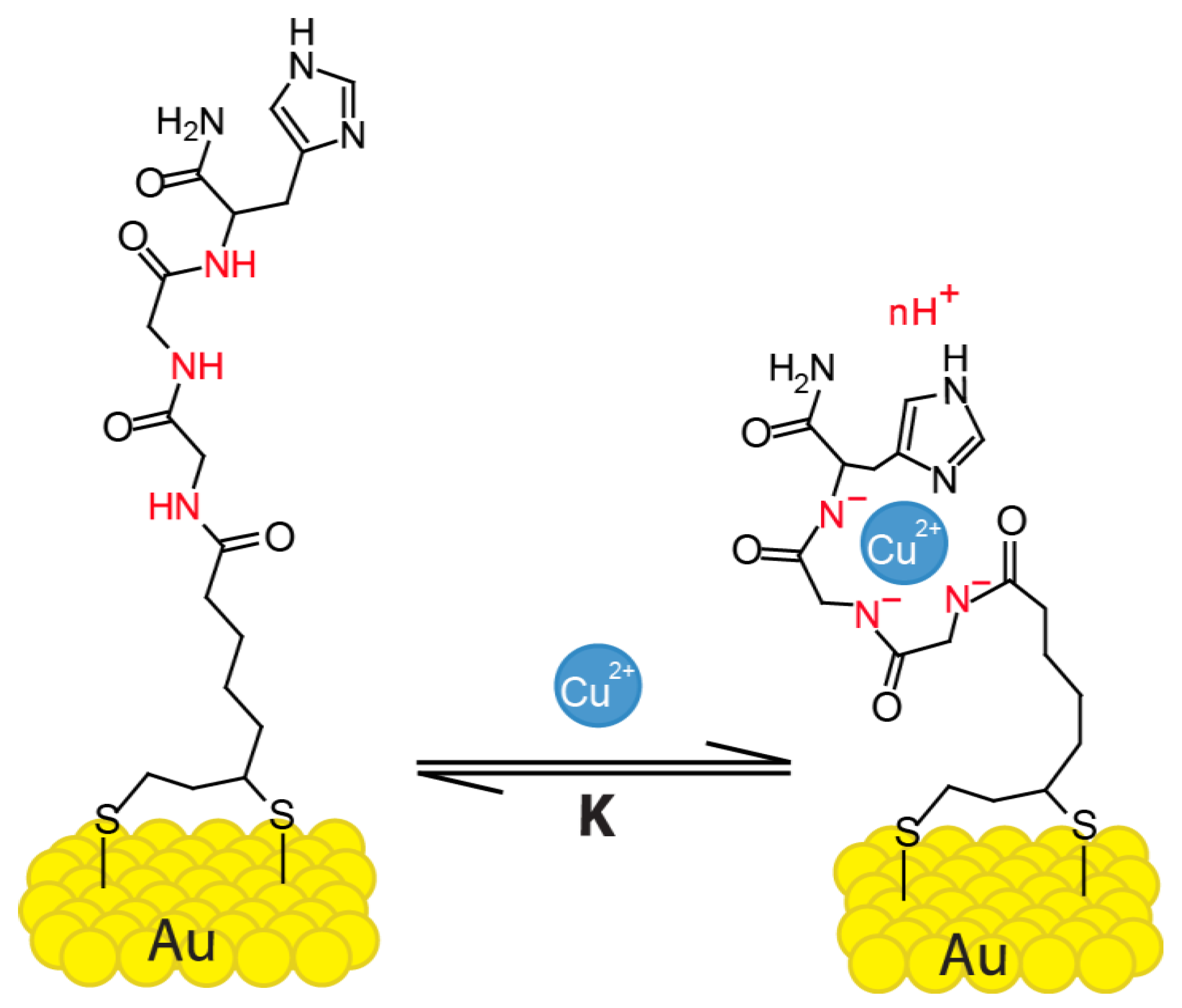
Why is Cu+ diamagnetic while Cu2+ is paramagnetic? | by KAKALI GHOSH , Teacher,blogger. M.Sc chemistry. | Medium

![For a complex ion, [Cu(NH3)4]^2 + : For a complex ion, [Cu(NH3)4]^2 + :](https://haygot.s3.amazonaws.com/questions/1731528_b11046ad9936443990f5efe5a262d1e8.png)