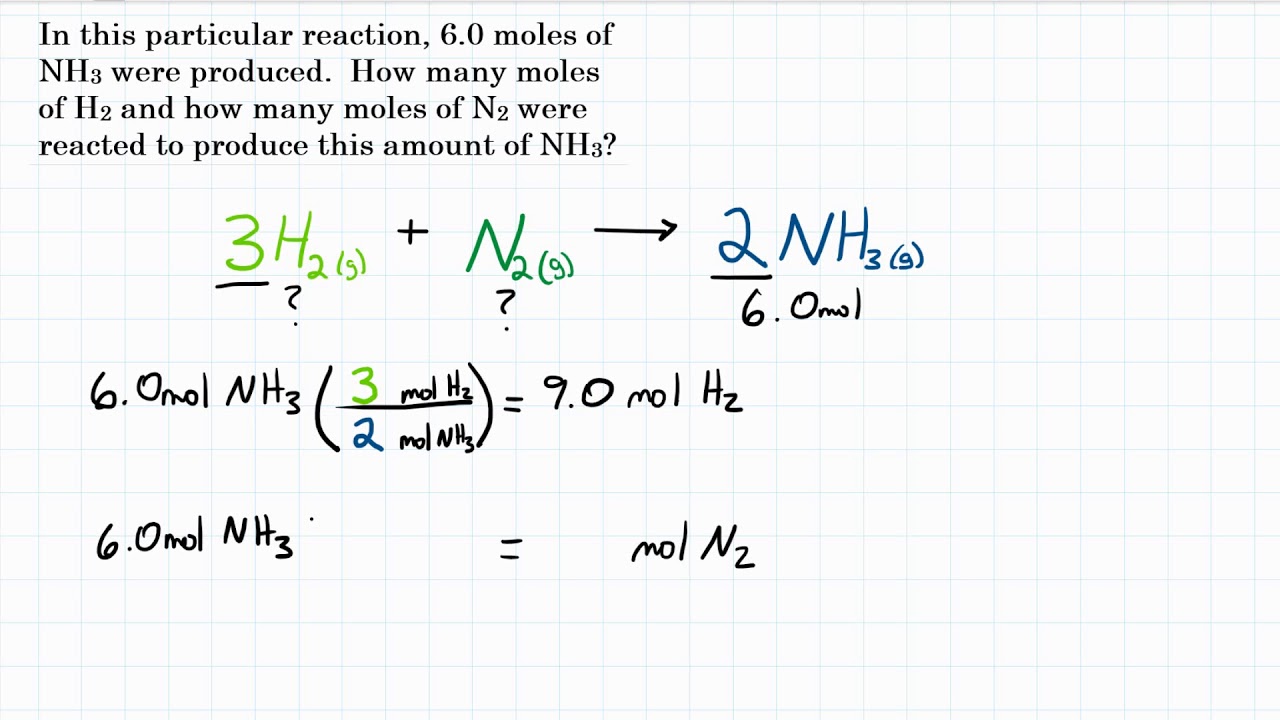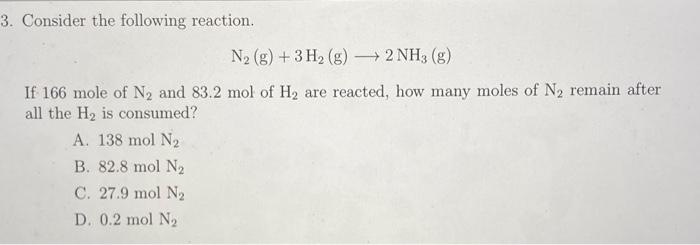One mole of N2 gas at 0.8 ATM takes 38 sec to diffuse through a pin hole, where as one mole of an unknown compound of Xenon with F at 1.6 ATM

⚗️PLEASE HELP!! DIMENSIONAL ANALYSIS AND MOLE CONVERSIONS CHEMISTRY!! Convert 0.30 moles of nitrogen - Brainly.com
When 1 mole of N2 and 1 mole of H2 is enclosed in 3 L vessel and the reaction is allowed to attain equillibrium ,it is found that at equillibrium there is

SOLVED: The reaction below is 0.20 mol of N2 and 0.90 mol of H2. How many moles of H2 gas will be left unreacted after the reaction has gone to completion? N2 (
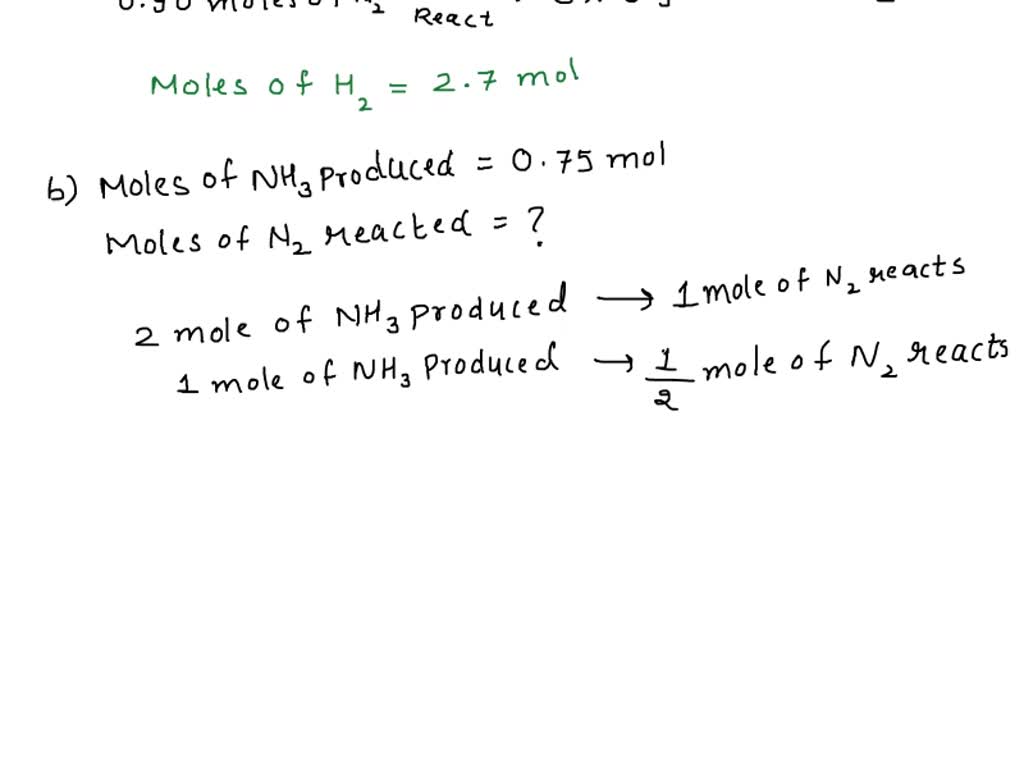
SOLVED: Ammonia is produced by the reaction of hydrogen and nitrogen as follows: N2(g)+3H2(g)→2NH3(g)ammonia A) How many moles of H2 are needed to react with 0.90 mol of N2? Express the number

SOLVED: How many moles of N2 are in a 500.0 mL container at 780 mm Hg and 135 oC? options: 0.0153 mol 0.0146 mol 0.440 mol 0.463 mol