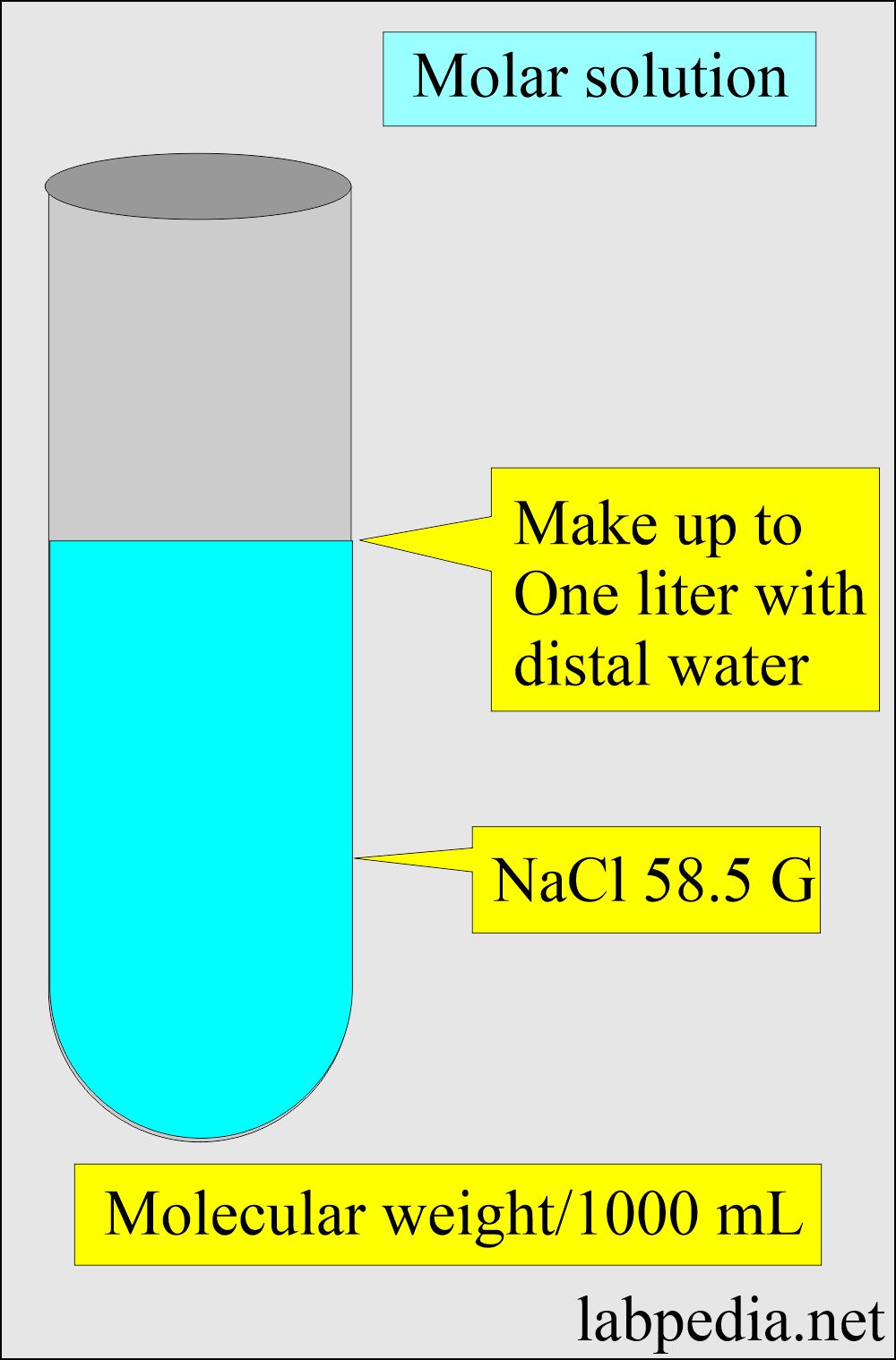
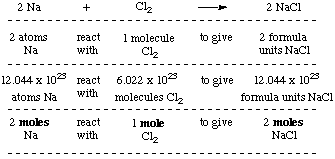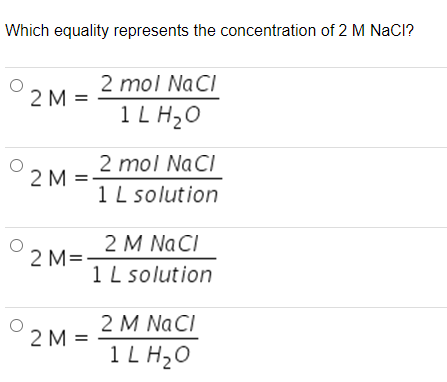The Mole & Chemical Quantities. The Mole Mole-the number of particles equal to the number of atoms in exactly 12.0 grams of carbon mol = 6.02 x. - ppt download

The density of `1 M` solution of `NaCl` is `1.0585 g mL^(-1)`. The molality of the solution is - YouTube

3/4/2016 I ObjectiveDo Now Convert grams of a substance to moles of a substance. Calculate the molar mass of: NaCl MgCl ppt download

1 Molar solution of sodium chloride | 1M solution of sodium chloride | 1 molar solution of NaCl - YouTube

Molarity Problems – IF worksheet. 1)MW NaCl = 23 + 35 = 58g NaCl = 1 mole 2)M = moles M = x moles Liters 1.0 L 3)58 g NaCl 1 mole NaCl = 1 mole NaCl 58. - ppt download

Calculate the mass of 1 mole of each one of the following: (a) `NaCl` , (b) `CaCO_(3)` , (c ) `FeSO - YouTube